Top 6 Publication Highlights from our Class 5 Photonics White Dwarf Users

We are excited to highlight some of the most prestigious publications by our White Dwarf 2P/3P laser users of Class 5 Photonics. Their groundbreaking neuroscience research continues to push the boundaries of scientific knowledge, earning recognition in top-tier journals. Here is our selection of the Top 6 publications:
1. Volatile Working Memory Representations Crystallize with Practice
Working memory is essential for cognitive functions, involving the temporary maintenance and manipulation of information. Working memory representations in the brain, particularly in the secondary motor cortex (M2), evolve and stabilize with practice. Initially, these representations are volatile and drift across days during the early stages of learning, but with continued practice, they become more stable, especially during the late-delay epoch of a working memory task. The study demonstrates that this stabilization is crucial for expert-level task performance. Through large-scale volumetric calcium imaging of up to 73,307 neurons at 6.5 Hz, the researchers observed that neurons in deeper layers of the M2 play a key role in maintaining stable working memory representations. This suggests that working memory circuits crystallize over time, leading to more efficient task performance as expertise develops.
Check the article here https://www.nature.com/articles/s41586-024-07425-w
|
Laser used: |
 |
White Dwarf 2p |
|
|
| Product Details |
2. High-speed, cortex-wide volumetric recording of neuroactivity at cellular resolution using light beads microscopy
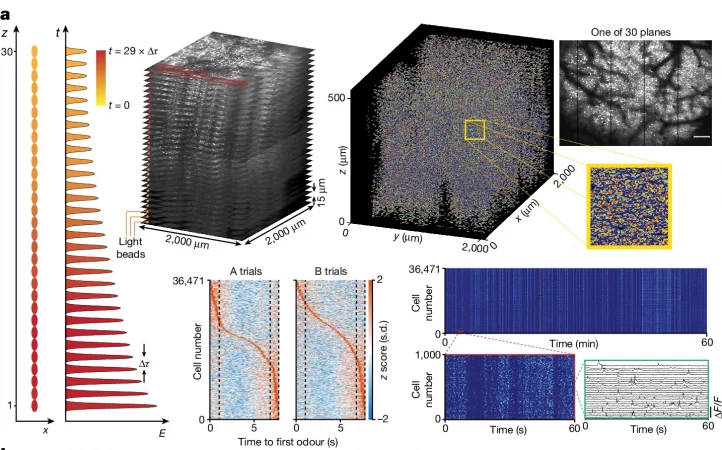
Two-photon (2P) microscopy allows high-resolution neuroactivity imaging in brain tissue but faces limitations in speed and spatiotemporal sampling for large-scale volumetric recordings. This study introduces light beads microscopy (LBM), an approach leveraging fluorescence lifetime to achieve scalable, high-speed volumetric recording at cellular resolution. LBM captures neuroactivity at 1.41 × 10^8 voxels per second, enabling mesoscopic imaging of the mouse cortex with detailed resolution. The technique demonstrates its potential by recording neuroactivity in volumes containing over 200,000 neurons at 5 Hz and up to 1 million neurons at 2 Hz, offering insights into cortex-wide information processing in the mammalian brain.
Check the article here https://www.nature.com/articles/s41592-021-01239-8

|
Laser used: |
 |
White Dwarf 2p |
|
|
| Product Details |
3. Volumetric Ca2+ Imaging in the Mouse Brain Using Hybrid Multiplexed Sculpted Light Microscopy
Calcium imaging with two-photon microscopy is vital in neuroscience, but traditional methods face trade-offs in field of view, speed, and depth. This study presents a new design paradigm using hybrid multi-photon acquisition for high-fidelity volumetric recordings at single-cell resolution within 1 × 1 × 1.22 mm volumes at up to 17 Hz in awake mice. The imaging system captures neuroactivity in the mouse auditory cortex, posterior parietal cortex, and hippocampus, demonstrating its ability to record up to 12,000 neurons, revealing the system’s versatility and potential for in-depth brain studies.
Check the article here https://www.cell.com/cell/fulltext/S0092-8674(19)30273-9
Video S2. Example 4× Axial Recording of Mouse neuronal activity. 3D rendering of a 30min volumetric recording in a volume of 690 × 675 × 600μm, 16.7Hz frame-rate, using a cytosolic GCaMP6f genetic calcium marker. The video shows the first 3min of the recording (Playback speed: 3 ×)
|
Laser used: |
 |
White Dwarf hybrid |
|
|
| Product Details |
4. High-resolution structural and functional deep brain imaging using adaptive optics three-photon microscopy
Multiphoton microscopy enables detailed visualization of neural cells and circuits but struggles with imaging performance at depth due to tissue scattering and optical aberrations. This study introduces a three-photon excitation methodology combined with adaptive optics (AO) and electrocardiogram gating for deep-tissue imaging. The approach achieves near-diffraction-limited imaging of cortical spines and dendrites up to 1.4 mm depth. Applications include deep-layer calcium imaging of astrocytes in highly scattering tissues, demonstrating significant advancements in intravital imaging capabilities.
Check the article here https://www.nature.com/articles/s41592-021-01257-6

|
Laser used: |
 |
White Dwarf 3p |
|
|
| Product Details |
5. A three-photon head-mounted microscope for imaging all layers of visual cortex in freely moving mice
Head-mounted microscopes have advanced neuronal activity imaging in freely moving mice but are typically limited to upper cortical layers and minimally lit environments. This study presents a lightweight, 2-gram, 3P excitation-based microscope with a z-drive for accessing all cortical layers in fully lit environments. The microscope’s on-board photon detectors are robust to environmental light, enabling functional imaging of cortical layer 4 and layer 6 neurons in various lighting conditions. The study shows differential modulation of neuronal activity in these layers under lit and dark conditions during free exploration.
Check the article here https://www.nature.com/articles/s41592-022-01688-9

|
Laser used: |
 |
White Dwarf 3p |
|
|
| Product Details |
6. The Cousa objective: a long-working distance air objective for multiphoton imaging in vivo
Multiphoton microscopy transforms neural imaging but is often limited by the mechanical and optical constraints of conventional objectives. This study introduces the Cousa objective, an ultra-long working distance (20 mm) air objective optimized for multiphoton imaging wavelengths. It offers a 4 mm² field of view with submicrometer resolution and is compatible with standard multiphoton systems. The novel design supports diverse in vivo imaging applications, demonstrated through successful imaging in various species, including nonhuman primates, enabling a broad range of neuroscience experiments.
Check the article here https://www.nature.com/articles/s41592-023-02098-1


Fig. Two-photon and three-photon imaging in mice. b, the mechanical design of the objective prioritized keeping the widest diameter near the middle of the objective to avoid mechanical collisions with objective mounts. All dimensions are in mm unless otherwise noted. Right: a photograph of the manufactured objective. c, Left: two-photon excitation PSF measurements were made with 0.2 µm beads embedded in agar at a depth of 350 µm. The excitation wavelength is 910 nm. z stack images are acquired for beads at four lateral locations including on axis, 1°, 2° and 3° off-axis (n = 5 beads at each location). FWHM of the Gaussian fits for measurements (mean values ± s.d.) indicate lateral and axial resolutions indistinguishable from diffraction-limited resolutions. The pixel size of the images is 0.058 × 0.064 × 0.69 µm3 (xyz). Right: images of a fluorescent calibration sample with a periodic line pattern (five lines per millimeter) in two orientations acquired under a ±5° scan angle show a nominal 2 × 2 mm2 FOV of the objective under the ±3° scan angle and a 3 × 3 mm2 FOV under ±5° scan angle. c, Left: the Cousa supports large FOV three-photon imaging, with a 20 mm WD. The vasculature across the entire 4 mm2 region is visible after an intravenous injection of Texas Red dextran. Right: higher zoom single z plane three-photon images from a second mouse with dual channel imaging of Texas Red dextran (magenta) and THG (cyan) in cortex and white matter. https://www.nature.com/articles/s41592-023-02098-1/figures/4 https://www.nature.com/articles/s41592-023-02098-1/figures/1
|
Laser used: |
 |
White Dwarf 3p |
|
|
| Product Details |
These publications underscore the exceptional research capabilities of our Class 5 Photonics White Dwarf customers. Their contributions to the scientific community are invaluable, driving innovation and expanding our understanding of complex biological processes. We are proud to support their endeavors and look forward to more groundbreaking discoveries in the future.
More about our 2-Photon and 3-Photon Microscopy Lasers



